New paper in Collaboration with Olivier Coulembier
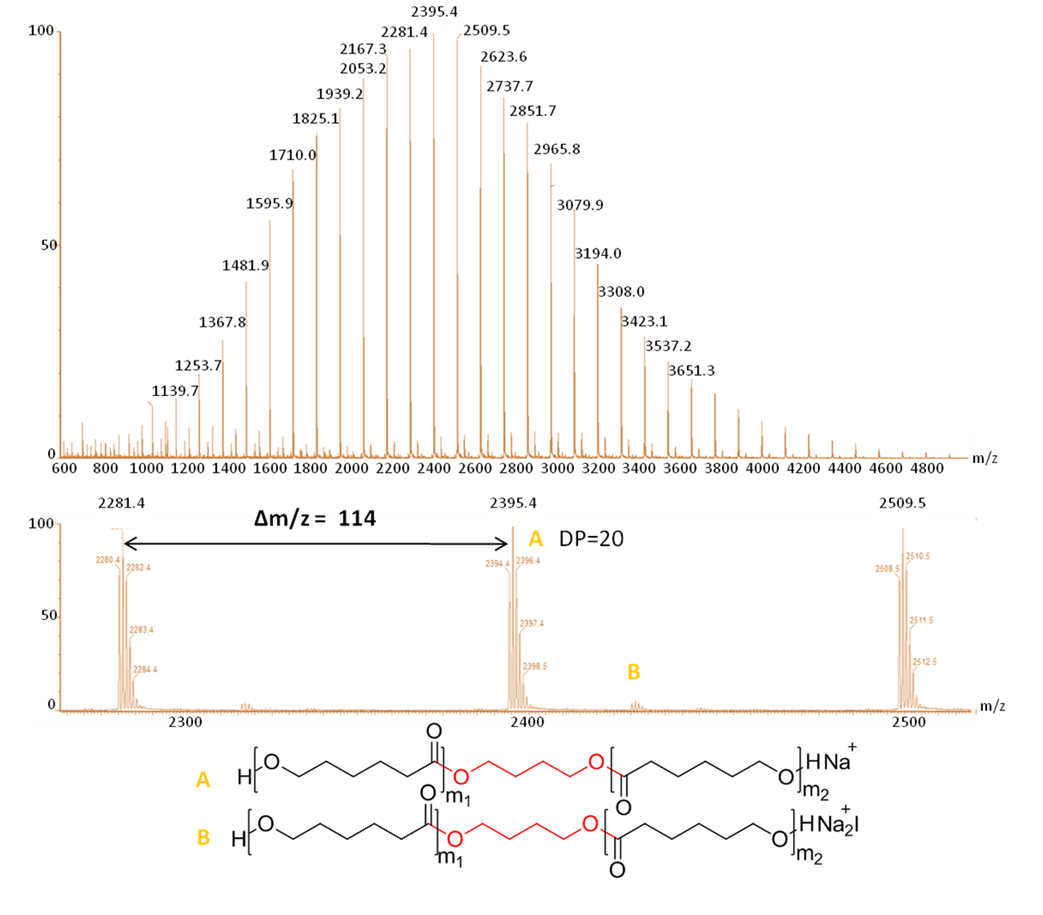
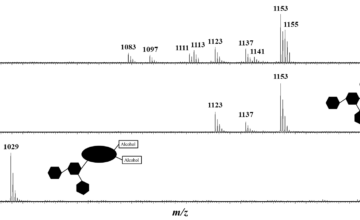
In this manuscript, a novel solvent-free and organocatalyzed ring-opening (co)polymerization (ORO(c)P) method utilizing benzoic acid(BA) as simple thermostable carboxylic acid-type catalyst is proposed to not only produce structurally well-defined aliphatic homopolyesters derived from L-lactide (L-LA) and Ɛ-caprolactone (CL), but also and, unexpectedly, statistical copolyesters based on the two monomer units. RO(c)P reactions were conducted in bulk in a temperature range of 155-180°C, in presence of alcohols as initiators. A triblock copolymer, namely, PLLA-b-PCL-b-PLLA, was also synthesized, attesting to the “controlled/living” character of this BA-OROP process. A bifunctional mechanism is proposed to operate, involving activation of both the monomer and the propagating hydroxyl by H-bonding. Very importantly, the BA organocatalyst could be readily recycled by simple sublimation and could be reused in further organocatalytic cycles. The perfect control over the mass parameters as well as the end-group fidelity were confirmed by mass spectrometry.