New paper in the collaboration with the Erasmus University Medical Center
A paper entitled “Identification of 4-Hydroxyproline at the Xaa Position in Collagen by Mass Spectrometry” has just been accepted for publication in the Journal of Proteome Research, an ACS scientific journal.
The paper concerns the identification of an enzymatic structural modification of the proline residues of collagen by mass spectrometry.
Collagen has a triple helix form, structured by a [-Gly-Xaa-Yaa-] repetition, where Xaa and Yaa are amino acids. This repeating unit can be post-translationally modified by enzymes, where proline is often hydroxylated into hydroxyproline (Hyp). Two Hyp isomers occur in collagen: 4-hydroxyproline (4Hyp) and 3-hydroxyproline (3Hyp).
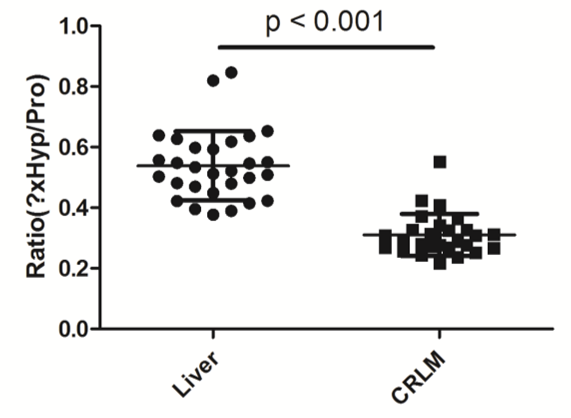
We showed that proline, at position 584 in COL1A2, has a lower rate of modification in colorectal liver metastasis tissue than in healthy liver, but the isomeric identity of ?xHyp, i.e. 3- and/or 4Hyp, remained unknown.
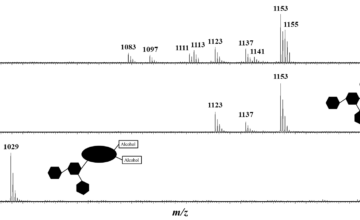
We here present a proof of principle identification of ?xHyp. This identification is based on liquid chromatography retention time differences and mass spectrometry using ETD-HCD fragmentation, complemented by ab initio calculations.
Both techniques identify ?xHyp at position 584 in COL1A2 as 4-hydroxyproline (4xHyp).